Content:
1. Clinical embryology of the eye
2. Histology of the eye and accessory organs
3. Physiology and metabolic processes of the visual perception
4. Metabolism of the eye tissues
_
Clinical embryology of eye
The most important structures of optical apparatus from the point of embryology involve neuroectoderm of prosencephalon, which gives rise to retina, nervus opticus and dorsal layer of cornea. Accommodative apparatus (lens and corneal epithelium) originates from superficial epithelium. Mesoderm gives rise to vascular and fibrous portion of eye and neural crest cells create origin for sclera and corneal endothelium.
The development of eye itself is relatively difficult process. Retina originates as an invagination of lateral side of prosencephalon right before closure of neural tube. It’s development is fastest first in area of eye pole. Macular area derives as soon as possible but after 4th month of prenatal development lags behind and its development starts again after birth until 4th month. Due to that the newborn is not able to see distinctly and to fix observed subject. The important myelination is finished in 10th postnatal week.
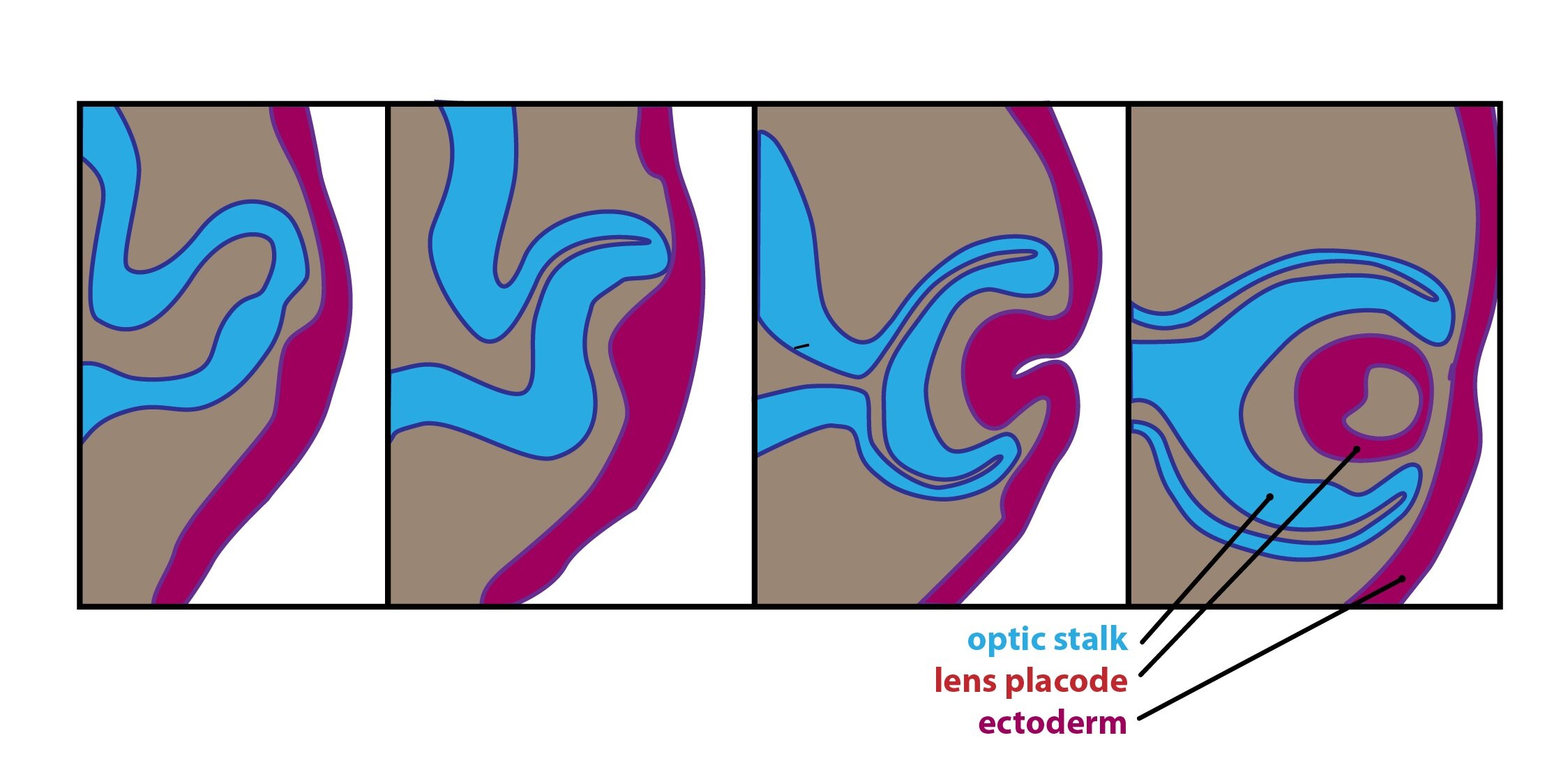
Development of eyeball is divided into several phases – optic groove, optic vesicles and stalk, lens placode, optic cups. The stage of optic cup is most important for development of congenital defects of eye. Invagination of optic vesicles creates double-layered optic cup. Invagination is asymmetrical and due to that is on the ventral side formed linear groove – fissura optica – necessary for connection between inner layer of optic cup and stalk. It contains mesenchyme, hyaloid artery and vein and nerve fibers of the optic nerve. Outer layer of optic cup is the outer layer of the pigment epithelium, whereas the inner layer is pars nervosa retinae.
Clinical correlations:
Congenital defects of eye are not so common but they represent the most often causes of blindness and vision defects in children. The most risk factor for developing eye is effect of teratogenes between 24th and 40th day of embryonal development. Defects can be genetically predisposed or caused by effect of exogenous infections. Follows several details about five most important congenital optic defects – coloboma, congenital glaucoma, kongenital cataract, microphtalmus and anophtalmus.
1) Colobom
This most common defect arise from incomplete closure of fisura optica and manifests as visible groove of corresponding structures. Defect is situated by the lower edge of iris which acquires the shape of key hole. Defect can be deeper and affect optic nerve, retina, choroid and ciliary body. This anomaly can be genetically predisposed or can be caused by exogenous effect e.g. rubeola or toxoplasmosis.
2) Congenital glaucoma
It is increase of intraocular pressure due to congenital defect in development of drainage of aqueous humor. Sinus venosus sclerae can be missing or filled with mesenchymal connective tissue. This defect can be caused by infection of rubeola in first trimester of pregnancy.
3) Congenital cataract
It is inherited, genetically conditioned cataract. Lens can be affected by rubeola infection and because of that is important to immunize young girls before they grow up to the reproductive age.
4) Microphthalmus
It is caused by abnormal development of optic vesicle and the eye is smaller. This defect can occur together with clefts of facial structures. Microphthalmus is genetically conditioned or caused by infectious agents (cytomegalovirus, toxoplasma gondii, rubeola virus, herpes simplex virus) which passes through the placenta.
5) Anophthalmus
Undeveloped lens, cornea and another structures of eye. This defect is commonly associated with craniocerebral malformations.
_
Histology of the eyeball and accessory structures of the eye
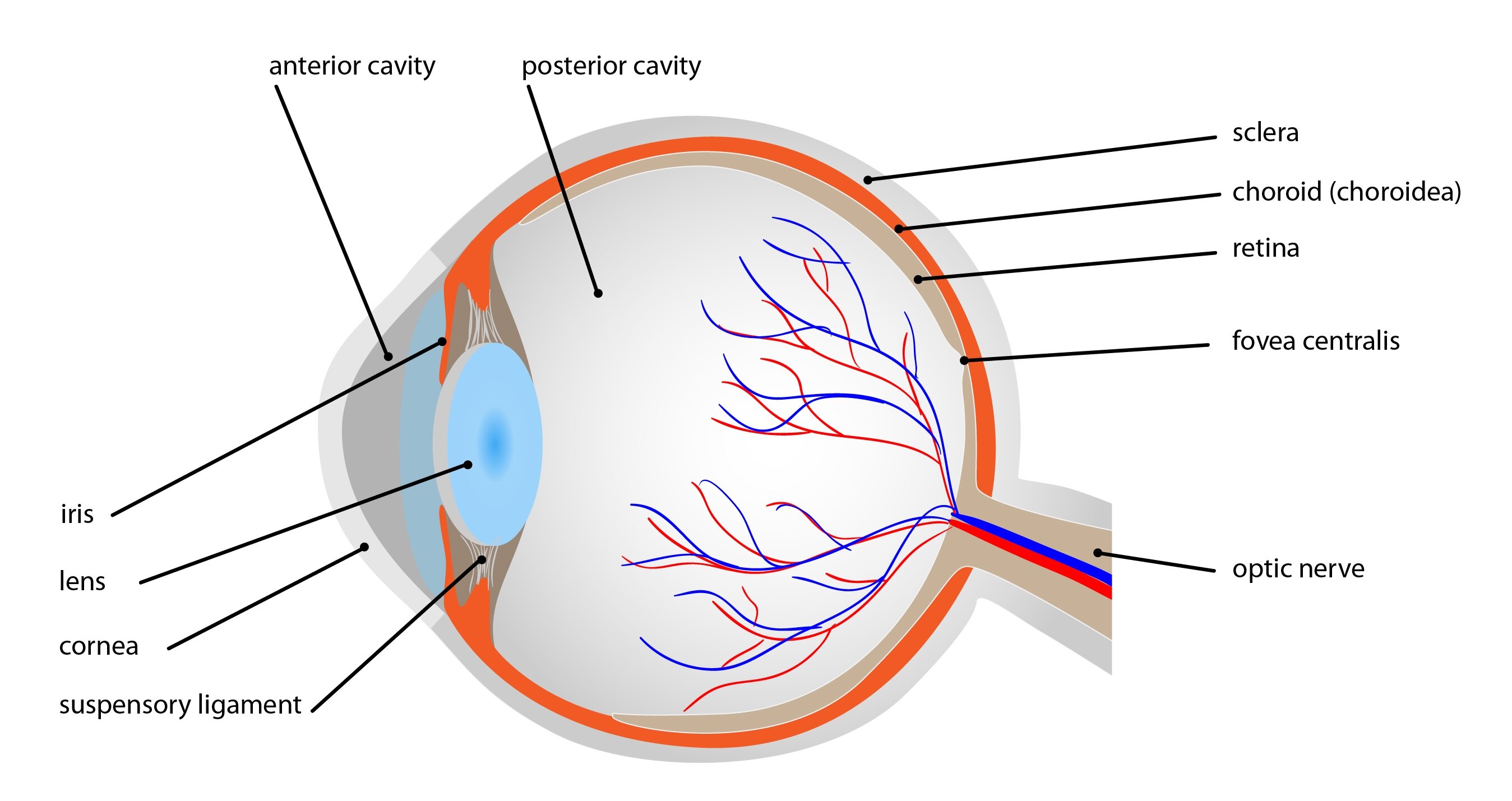
The wall of the eye consists of three layers:
1) Tunica fibrosa (outer) – sclera, cornea
2) Tunica vasculosa (middle) – uvea, ciliary body, iris
3) Tunica nervosa (inner) – retina
And three chambers: camera oculi anterior (anterior chamber) a camera oculi posterior (posterior chamber) filled with fluid and corpus vitreum (vitreous chamber).
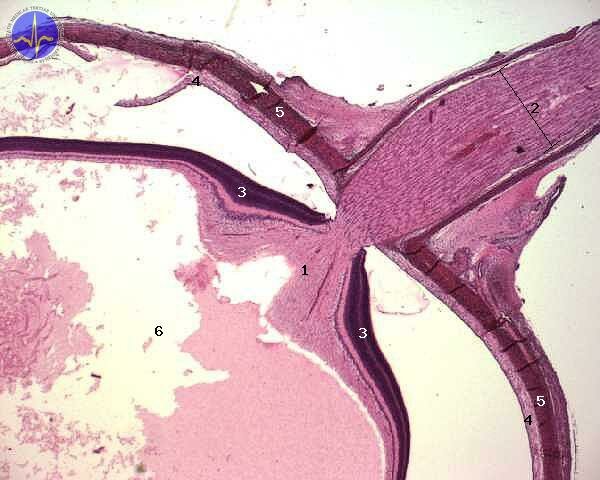
1 – Discus nervi optici, 2 – Nervus opticus, 3 – Retina, 4 – Choroidea, 5 – Sclera, 6 – Camera of vitreal body
Follows the detailed description of the layers:.
1) Tunica fibrosa
It is outer fibrous layer including sclera and cornea. Sclera is the white avascular layer of dense collagen bundles with small amount of fibroblasts. Cornea is transparent structure representing the ventral part of tunica fibrosa. It is composed of 5 layers: corneal epithelium, Bowman’s membrane, corneal stroma, Descemet’s membrane, corneal endothelium.
The corneal epithelium is nonkeratinized stratified squamous consisting of 5-6 cellular layers with lipids and glycoproteins. The corneal epithelium has regenerative ability.
Bowman’s membrane is composed of irregularly arranged collagen fibers and it ensures stability and resistance of cornea.
Stroma is avascular tissue consisting of flattened fibroblasts and extracellular matter with proteoglycans rich in chondroitin sulfate.
The Descemet’s membrane holds the post of basal membrane.
The corneal endothelium is formed by one layer of flattened cells with ability of proteosynthesis. It contains only minimum of fluid and ensures corneal transparency.
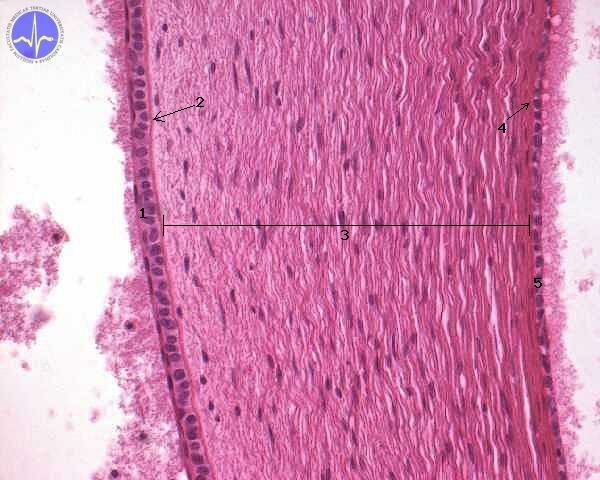
1 – Anterior epithelium of cornea, 2 – Lamina limitans anterior (Bowman’s membrane), 3 – Stroma of the cornea, 4 – Lamina limitans posterior (Descement’s membrane), 5 – Posterior epithelium of cornea
2) Tunica vasculosa
This middle layer is composed of choroid (choroidea), ciliary body (corpus ciliare) and iris (iris). Highly vascularized choroid contains loose collagenous connective tissue with fibroblasts, macrophages, mast cells and plasma cells. It consists of 4 layers: lamina suprechoroidea, zona vasculosa, lamina choriocapillaris, lamina vitrea – Bruch’s membrane.
Ciliary body is covered by pars ciliaris retinae (ciliary part of retina), whose outer layer contains pigmented epithelium and inner layer contains non-pigmented epithelium.
Iris partially covers the lens as projections of choroid. The central aperture creates pupil.
The amount of light entering the eye is regulated by increase or decrease of pupil size. These changes are under control of sympathetic and parasympathetic innervation. The parasympathetic nerve fibers begin in nucleus oculomotorius accesorius dorsalis Edinger-Westphal and continue together with fibers of nervus oculomotorius via fissura orbitalis superior. They enter the orbit into ganglion ciliare. Postganglionic fibers nervii breves penetrate the dorsal optic pole to reach musculus sphincter pupilae and musculus ciliaris. Sympathetic fibers are not crossed, they descend from hypothalamus into ganglion cervicale superius. Postganglionic fibers continue along arteria carotis to ganglion trigeminale and with ophthalmic nerve into musculus dilatator pupillae.
3) Tunica nervosa
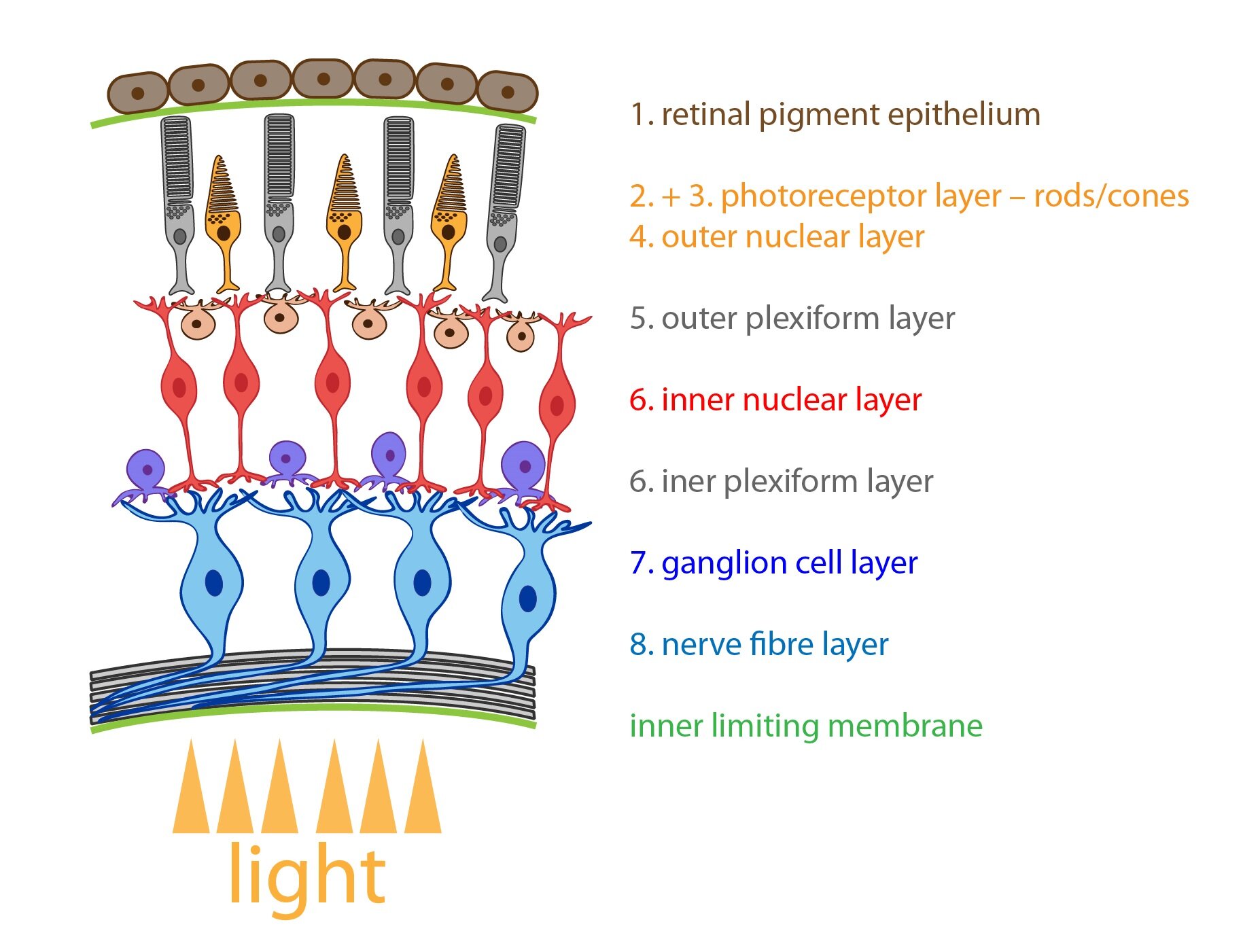
It is inner layer which creates retina. Retina lines the inner surface of eyeball. It composes of pars optica retinae (pigment epithelium + nerve tissue) and pars caeca retinae. The picture with description of individual structures follows below.
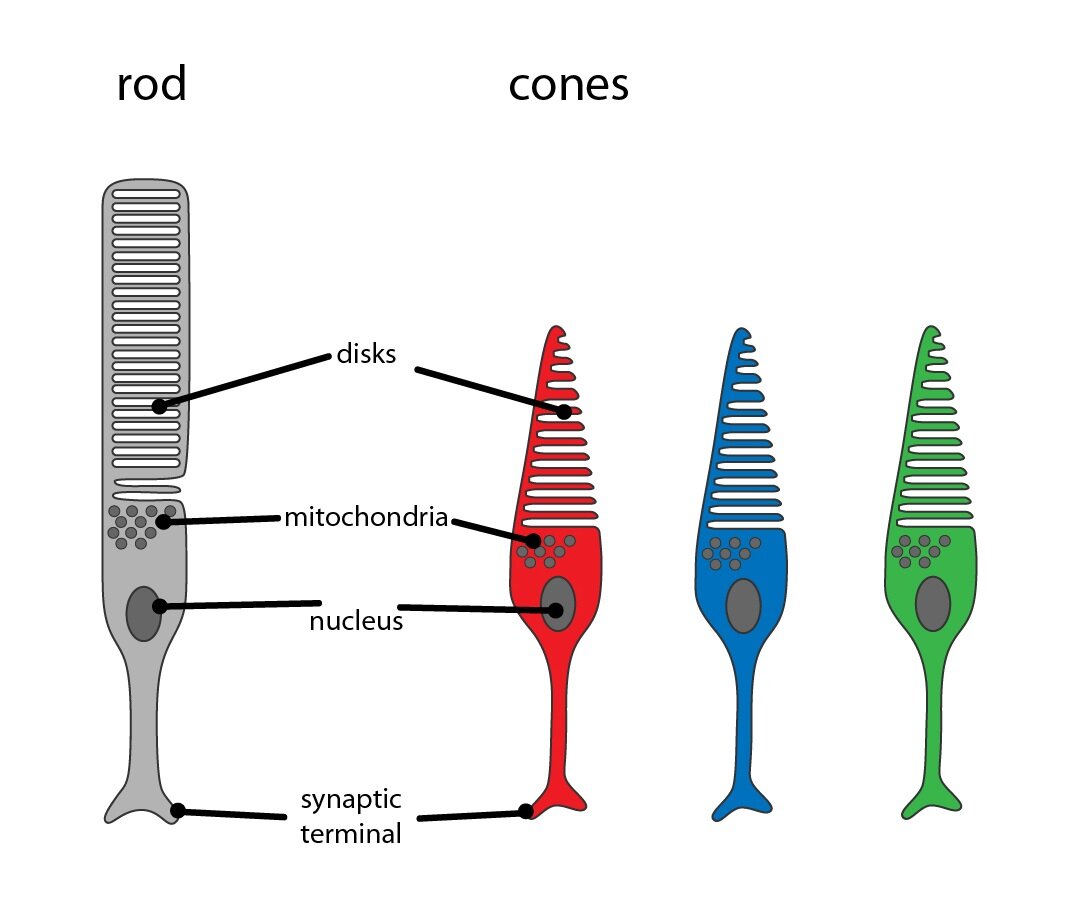
1. pigment epithelium – columnar epithelium with numerous microvilli (form columnar sheaths surrounding and dividing rods and cones). At the basal portion is located basal labyrinth ensuring the ion transport. The apical portion is filled with melanin granules.
2. Photoreceptor cells – rods and cones – are primary receptor cells or more precisely modified bipolar neurons. Retina contains about 120 million rods, they are composed of outer photosensitive segment and inner segment that ensures protein synthesis. They contain visual pigment rhodopsin which is disintegrated by the effect of light into opsin and retinal (vitamin A aldehyde). That is the reason why deficiency of vitamin A causes deterioration of vision in dim light – hemeralopia. Cones (B) have similar structure as rods but they differ in size and bottle shape. In their membranes is involved iodopsin which enables colour vision. The colour vision is in the human eye localized into 3 types of cones which are able to absorb the light of different wavelength: blue (maximum absorbance 420 nm), green (maximum absorbance 535 nm) and red (maximum absorbance 565 nm). Human vision is normally trichromatic – thus, all colours are composed of these three basic colours. There are some pathological conditions when deficit of colour perception occurs especially of green and red colour which causes colour blindness (daltonism).
3. Outer limiting membrane
4. Outer nuclear layer
5. Outer plexiform layer
6. Inner nuclear layer
7. Inner plexiform layer
8. Ganglion cell layer
9. Layer of optic nerve fibers
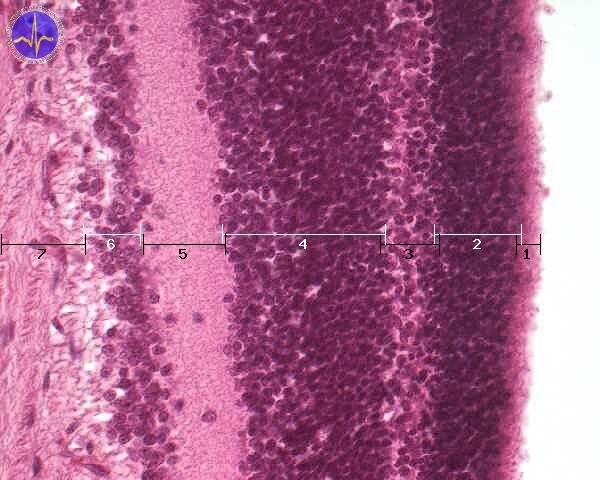
1 – Outer segments of cones and rods, 2 – Outer nuclear layer (nucleuses of rods and cones), 3 – Outer plexiform layer, 4 – Inner nuclear layer (nucleuses of bipolar, amacrine and horizontal cells), 5 – Inner plexiform layer, 6 – Ganglious cell layer, 7 – Optic nerve layer
Refractory structures of the eye
The refractory structures include cornea, lens, aqueous humor and corpus vitreum (vitreous body).
Lens is transparent highly elastic biconcave structure. I tone of the most important refractory organs. It bends light rays. It composes of 3 parts – lens capsule, subcapsular epithelium and lens fibers arranged into hexagonal prism.Capsula lentis contains glycoproteins, type IV. collagen and surrounds the whole lens. Its ventral part is disrupted during cataract surgery to implant the synthetic lens. Subcapsular epithelium on the ventral side is simple cuboidal.
Vitreous body fills the vitreous chamber. It contains 99 % of water, collagen and hyaluronic acid.
Accessory structures of the eye
The accessory structures include conjunctiva, eyelids and lacrimal apparatus.
Conjunctiva is composed of stratified columnar.
Palpebrae (eyelids) contain 3 types of glands. Meibomian glands (tarsal glands) are branched alveolar sebaceous glands which empty into conjunctival fornix. Their secretion prevents to excessive evaporation of tears. Smaller glands of Zeiss are branched sebaceous glands of eyelashes and empty into hair follicles of eyelashes. Glands of Moll are simple tubular apocrine glands and their ducts open into hair follicles of eyelashes.
The lacrimal apparatus (apparatus lacrimalis) is distinguished into two parts – glandular systém and ductal systém. Glandular system consists of lacrimal gland (glandula lacrimalis) and from accessory lacrimal glands in eyelids and conjunctiva. This system produces tears into lacrimal sac. The ductal system transports tears to the nasal cavity. It begins with blind-ended lacrimal puncta (punctum lacrimale) which continue to the lacrimal canaliculi (canaliculi lacrimales, stratified squamous epithelium), lacrimal sac, stratified ciliated columnar epithelium) and ends as nasolacrimal duct (ductus nasolacrimalis).
_
Physiology and metabolic processes of the visual perception
Visual perception is the most important sense human poses. About 80 % information of our surroundings is gained through it. We are able to distinct between two object, judge their qualities and their mutual distance. These abilities are essential for spatial orientation.
Sun beams enter the eye through different optical environments: cornea, anterior chamber, lens and vitreous body. Image is formed on retina. Due to physical properties of optical apparatus, image is smaller and flipped upside down.
Visual perceptions are leaded through four neuronal pathway from retina to visual cortex in occipital lobe.
1) First neuron is a photoreceptor, which can be found in neuroepithelial stratum of retina. There are two kinds of photoreceptors: rods for visual perception in dark environment and cones for colour distinction. Rods and cones detect light beams. Photons evoke an action potential, that is transmitted to the second neuron.
2) Second neuron is bipolar cell, which originates in retinal ganglion. Some of them collects information from rods, some of them from cones.
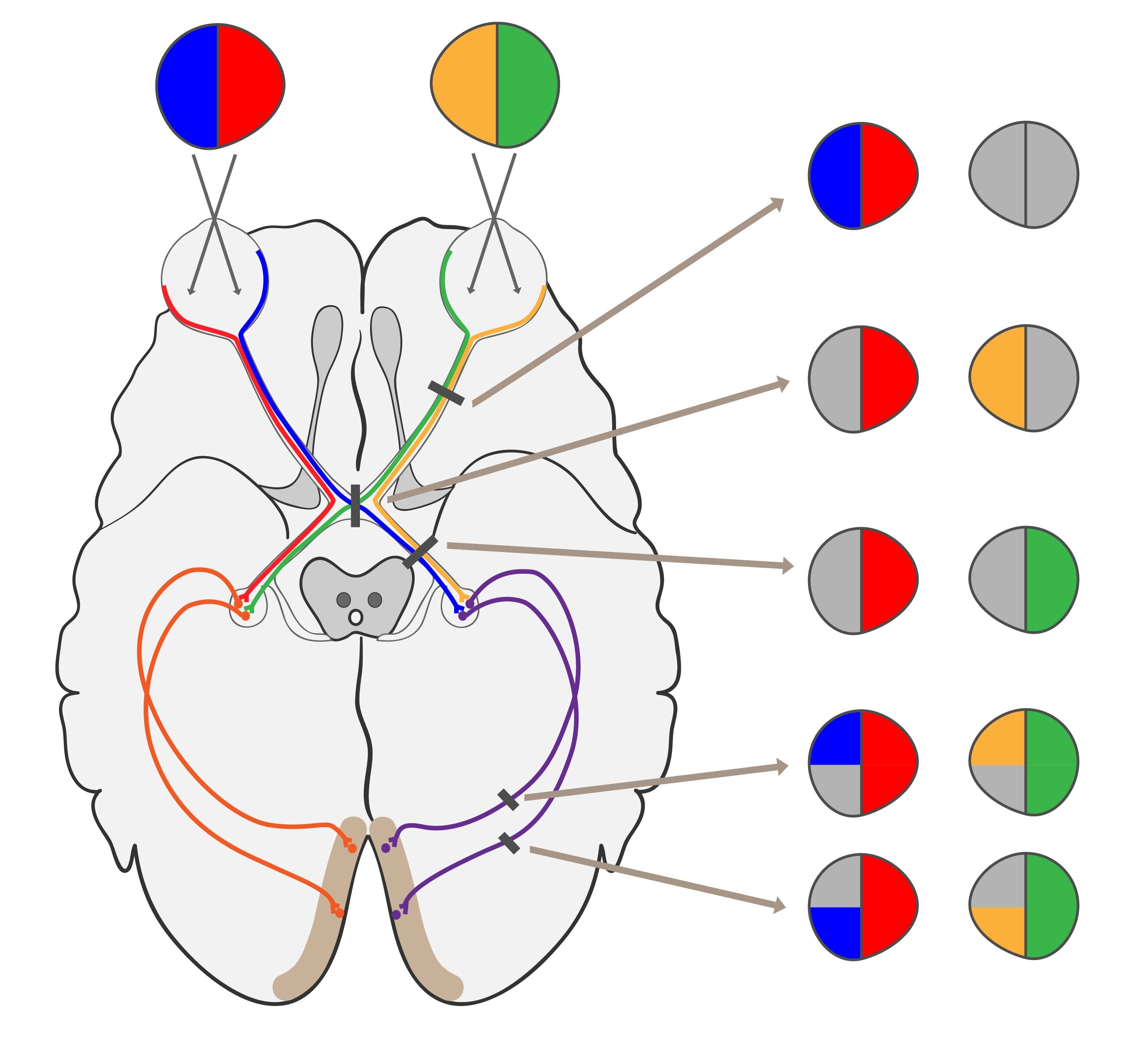
3) Third neuron is a multipolar cell with perikaryon on the inner surface of the retina in so called optic nerve ganglion. Axons of these multipolar neurons form optic nerve. It is said that left and right optic nerve crosses in optic chiasm. In fact about half fibres remain on the ipsilateral side and do not cross at all. On the other hand nasal portion of fibres truly cross. Continuation of visual pathway is called optic tract past optic chiasm. Axons forming an optic tract synapse in lateral geniculate nuclei.
4) Fourth neuron can be found in lateral geniculate nuclei. Its projection continues to visual cortex.
Impairments of specific level have specific signs, which are important to distinguish:
|
Lesion |
Sign |
| Prior to optic chiasm | Complete loss of ipsilateral visual field |
| In proper optic chiasm | Bitemporal hemianopsia with loss of both temporal halves of visual field (due to sclerosis of carotid artery) Binasal hemianopsia (due to hypophyseal tumor) |
| Between optic chiasm and lateral geniculate corpus | Homonymous anopsia |
| Post lateral geniculate corpus | Quadrantal hemianopsia |
_
Mechanism of photon reception
Rods function lies as nocturnal vision receptors. Cones empower our sight to differ various colours. Rods of human eye contains a photon sensitive pigment rhodopsin. Rhodopsin reacts with photons and breaks down to opsin (proteinic part) and retinal (non proteinic part). Retinal is derived from vitamin A (chemically it is an aldehyde of vitamin A). This is also a reason why a low vitamin A intake cause visual impairment in dark environment – hemeralopia. Synthesis of retinal from vitamin A is a key to proper photo adaptation to different luminous conditions.
Mutation in a rhodopsin gene causes an inherited degenerative disease called retinitis pigmentosa, which can lead to a complete loss of sight. Rhodopsin can be found in a rods membranes, where forms up to 85 % of all proteins. In fact rhodopsin is a receptor coupled with G – protein (rods G – protein is called transducin).
Photon activates rhodopsin, which activates transducin. Cascade continues by activation of phosphodiesterase (PDE). PDE catalyses hydrolysis of cGMP to non cyclic GMP. With decrease in concentration of cGMP sodium and calcium channels close, which leads to hyperpolarization. Hyperpolarisation spreads to the first synapse of visual pathway. Transport of sodium to extracellular compartment is mediated by Na+/K+-ATPase. This cascade as a whole is called vertebrate phototransduction cascade.
Colour vision
Colour vision is achieved by three distinct kinds of cones. They differ in a colour of light absorbing pigment:
1) Blue pigment – absorption maximum 420 nm
2) Green pigment – absorption maximum 535 nm
3) Red pigment – absorption maximum 565 nm
So that colour vision is trichromatic in humans. All the others colours are composed from these three (trichromatic theory: Young-Helmholtz, 1872). Differences in the absorption maximums are caused by protein part of pigment. 11-cis -retinal is the same retinal molecule, as it is in the rods. There is about 1-2% of population with impaired colour vision due to defect of a photoreceptive protein. The most common are impairments in distinction of green or red – this pathologic state is called daltonism. Men are commonly afflicted because colour blindness is X-bounded impairment.
Mechanism of action potential generation remains the same in cones as it was in rods.
Molecular changes in rhodopsin
Rhodopsin is a complex of opsin and 11-cis-retinal. Photon causes isomerization of 11-cis-retinal. Product is called all-trans-retinal. This process causes conformational changes in rhodopsin as is truth for rods or colour pigments in cones. All-trans-retinal detaches itself when isomerization is completed.
Photon energy cause 11-cis-retinal to undergo several conformational changes: Bathorhodopsin to lumirhodopsin, lumirhodopsin to metarhodopsin I and at last: metarhodopsin I to metarhodopsin II, which activates the transducin. Metarhodopsin II dissociates to opsin and all-trans-retinal immediately after transducin activation. All-trans-retinal is converted to all-trans-retinol by all-trans-retinal dehydrogenase. All-trans-retinol is then transported to pigmented epithelium, where it is converted to 11-cis-retinol. 11-cis-retinol is transported back to the outer segment of the rod and re-oxidise to 11-cis-retinal. Finally, opsin reacts as Schiff’s base with retinal aldehydic group and forms rhodopsin. The cycle is closed.
Cascade termination and regeneration
Termination of cascade is possible due to three basics processes:
1) Transducin (its α-subunit) spontaneously has a GTPase activity. It means, that it is entirely possible for transducin to hydrolyse GTP to GDP and turn itself off by doing so. This phenomenon is shared by all G-proteins.
2) Rhodopsin kinase can bind activated rhodopsin (Metarhodopsin II) and competes with transducin for binding site. While bound to rhodopsin, it phosphorylates opsin. So the affinity to transducin is lowered. On the other hand affinity to arestin is increased. Arestin prevents binding of transducin to rhodopsin. The dissociation of all-trans-retinal follows.
3) Calcium ions enter the rod cytoplasm through calcium channel in darkness. Light causes calcium channels to close, so the influx ceases. 3Na+/Ca2+-transporter further decreases intracellular calcium concentration. This process activates guanylate cyclase, which creates cGMP, cGMP acts as an activator of sodium and calcium channels. With channels opened, rod is prepared to register another photon.
_
Metabolism of the eye tissues
Main metabolic substrate is a glucose, as is true for the other parts of central nervous system.
Metabolism of cornea, tear compounds and aqueous humour
Tears
Outer surface of the cornea is exposed to a changing environment. Therefore cornea is protected by a tears, which are constituting three basic layers.
There is an oil coat in contact with the air. Oil coat is produced by a glands of Meibom. Its compounds are triacylglycerol, free fatty acids and cholesterol. The main purpose of the oil coat is to slow down evaporation of a watery tears compound below. It also strengthens a surface tension, so that tears do not spill over the operculum.
95 % of middle watery coat volume is produced by lacrimal gland. The rest is produced in glands of Wolfring and Krause.
Middle layer is 99 % a water. Another compounds are: glucose, vitamins, minerals, amino acids, enzymes, proteins, lactoferrin, lysozyme, immunoglobulins (IgA, IgE). As you can see from its compounds, there are some effective antibacterial substances, which protect the eye. Middle layer also improves oxygen transport and optical attributes of the cornea.
There is a mucous coat right next to corneal epithelium. Mucous coat is produced by goblet cells. This layer changes a hydrophobic nature of epithelial surface to the hydrophilic. Which facilitates its washing by the middle layer.
According to some authors there are no three layers (or coats if you like it). They suggest that these layers are mixed up.
Tear function review:
1) Optimisation of corneal optical properties
2) Scavenging bacteria and impurities
3) Keeping high level of humidity
4) Facilitating corneal nutrition
5) Eye protection from from infections (lysozyme, lactoferrin, immunoglobulins, etc.)
Formation, composition and drainage of aqueous humour
Inner surface of cornea is washed by a aqueous humour. It ensures nutrition for cornea as well as for lens. Aqueous humour is composed of water (99 %), it is colourless liquid containing amino acids, minerals, proteins, ions, lactate and vitamin C. Production of aqueous humour is continuous and takes place in prominences of ciliary body.
Its drainage is provided by following ways:
1) Pathway through trabecular meshwork into the Schlemm’s canal
Aqueous humour flows from posterior chamber through the pupil to the anterior chamber and from there, it exits the eye through the trabecular meshwork into the Schlemm’s canal, which is located at the joining point of the cornea and sclera. And finally through Schlemm’s canal to episcleral veins.
2) Uveoscleral drainage
This drainage leads through ciliary muscle to the supraciliar and suprachoroidal spaces.
3) Iris resorption (minimal participation)
Intraocular pressure is mainly determined by the coupling of the production of aqueous humour and it drainage. Any drainage impairment leads to increase in the intraocular pressure. This process leads to optic nerve damage and occurrence of a glaucoma.
Inner corneal surface is covered with polygonal endothelial cells. Every newborn has about 4000 – 5000 cells per square millimetre. The number of cell decreases to only 2000 cells per square millimetre during his 60th. These endothelial cells don’t undergo mitosis at all. If the endothelium is damaged cells are fixing the lesion by increasing their volume and surface. Their function is maintaining proper hydration by removing excess sodium using Na+/K+-ATPase. If there is less than 500 endothelial cells, stromal oedema appears.
Nutrition and oxygenation of cornea
There are three main sources of the oxygen:
1) Through tears from the atmospheric oxygen – only effective with the eyes open.
2) From aqueous humour – main source for endothelial cells
3) From conjunctival arteries – main source with the eye closed
Sources of glucose are the aqueous humour and tears. 30 % of glucose is utilised by the means of glycolysis, 65 % by the means of pentose cycle. NADPH produced by pentose cycle is used by corneal glutathione reductase.
Composition, metabolism of vitreous humour and lens
Vitreous humour is a gelatinous matter rich on collagen. Its main function is to maintain eyes shape and volume. It is formed by delicate collagen fibres with molecules of hyaluronic acid attached. Hyaluronic acid binds water molecules, that form a 99 % of vitreous humour volume. There are some fibrocytes and lymphocytes at the periphery of the vitreous humour. Cellular component helps to maintain collagen fibre and hyaluronic acid molecules.
There is no vessels in the lens. Its metabolic demands are satisfied by the aqueous humour. Lens consists of proteins and water. The most important proteins are α-, β- a γ-crystallins, that have to be kept in pure crystalline form. Osmotic balance is maintained by activity of a Na+/K+-ATPase. Free radicals are scavenged by glutathione reductase. Glucose is utilised by following way: 85 % glycolysis, 10 % pentose cycle, 3 % Krebs cycle.
Subchapter Authors: Martina Šajdíková, Patrik Maďa and Josef Fontana